دورة المؤشرنت للتحليل الفني
50 دينار كويتي
أنت تستخدم أحد المتصفحات القديمة. قد لا يتم عرض هذا الموقع أو المواقع الأخرى بشكل صحيح.
يجب عليك ترقية متصفحك أو استخدام أحد المتصفحات البديلة.
يجب عليك ترقية متصفحك أو استخدام أحد المتصفحات البديلة.
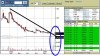
سهم لقطة حللوه وشوفوه
- بادئ الموضوع ممدوح
- تاريخ البدء
حبيب القمر
عضو نشط
والله انك طيب ياممدوح نصايحك طيبه مثلك
ياطيب القلب ياممدوح
جزاك الله خير اللهم ارحم والديك
عشت ياطيب
ياطيب القلب ياممدوح
جزاك الله خير اللهم ارحم والديك
عشت ياطيب
doji
عضو نشط
- التسجيل
- 31 أغسطس 2002
- المشاركات
- 1,136
عزيزي ممدوح تحية من الاعماق
السلام عليكم ورحمة الله وبركاته
عزيزي الفاضل ممدوح دائما وابداً اسهمك لا يختلف عليها اثنين وهذا يدل على قدرتك على اختيار افضل الاسهم وهذه صفه قد لا تتوفر الا في مضارب محترف .
ودائما تشير الى هذه الاسهم الى زملائك وهذا دليل على معدنك الطيب .
عزيزي الفاضل تستحق الاحترام كل الاحترام ولكن هذا السهم لايستحقه .
من ترك شيئاً لله عوضه بافضل منه او كما قال صلى الله عليه وسلم
تمنياتي القلبية لك بالتوفيق والسداد
تحياتي للجميع
السلام عليكم ورحمة الله وبركاته
عزيزي الفاضل ممدوح دائما وابداً اسهمك لا يختلف عليها اثنين وهذا يدل على قدرتك على اختيار افضل الاسهم وهذه صفه قد لا تتوفر الا في مضارب محترف .
ودائما تشير الى هذه الاسهم الى زملائك وهذا دليل على معدنك الطيب .
عزيزي الفاضل تستحق الاحترام كل الاحترام ولكن هذا السهم لايستحقه .
من ترك شيئاً لله عوضه بافضل منه او كما قال صلى الله عليه وسلم
تمنياتي القلبية لك بالتوفيق والسداد
تحياتي للجميع
الملفات المرفقه:
خبر جيد high.30
Wednesday, August 06, 2003 08:30 ET
CANTON, Mass., Aug 6, 2003 /PRNewswire-FirstCall via COMTEX/ -- Avitar, Inc. (Amex: AVR) announced today that it has signed a Letter of Understanding with Immunomatrix, Inc., for the exclusive right to utilize Immunomatrix's high sensitivity chemical assay technologies in Avitar's rapid, oral fluid drugs- of-abuse ORALscreen(TM) product line. The terms were not disclosed and are subject to the completion of a Definitive Agreement.
"We are very excited about the collaboration with Immunomatrix," said Peter P. Phildius, Chairman of the Board. "Their technology will accelerate our ability to deliver to the marketplace the next generation of oral fluid drugs-of-abuse testing by enabling us to produce a point-of-care test with enhanced sensitivity."
"Combining our technology expertise with Avitar's leadership in the rapid, oral fluid drugs-of-abuse market affords Immunomatrix another opportunity to provide the diagnostic community with novel, proprietary technology," said Allan M. Weinstein, M.D., Chairman & Chief Executive Officer of Immunomatrix. "We look forward to working with the Avitar team in this development project."
Avitar, Inc. develops, manufactures and markets innovative and proprietary products in the oral fluid diagnostic market, disease and clinical testing market, and customized polyurethane applications used in the wound dressing industry. Oral fluid diagnostics include the estimated $1.5 billion drugs-of- abuse testing market, which encompasses the corporate workplace and criminal justice markets. Avitar's products include ORALscreen(TM), the world's first non-invasive, rapid, onsite oral fluid test for drugs-of-abuse, and HAIRscreen(TM), a laboratory-based hair test for detecting long-term drug abuse. Additionally, Avitar manufactures and markets HYDRASORB(TM), an absorbent topical dressing for moderate to heavy exudating wounds. For more information, see Avitar's website at www.avitarinc.com.
Wednesday, August 06, 2003 08:30 ET
CANTON, Mass., Aug 6, 2003 /PRNewswire-FirstCall via COMTEX/ -- Avitar, Inc. (Amex: AVR) announced today that it has signed a Letter of Understanding with Immunomatrix, Inc., for the exclusive right to utilize Immunomatrix's high sensitivity chemical assay technologies in Avitar's rapid, oral fluid drugs- of-abuse ORALscreen(TM) product line. The terms were not disclosed and are subject to the completion of a Definitive Agreement.
"We are very excited about the collaboration with Immunomatrix," said Peter P. Phildius, Chairman of the Board. "Their technology will accelerate our ability to deliver to the marketplace the next generation of oral fluid drugs-of-abuse testing by enabling us to produce a point-of-care test with enhanced sensitivity."
"Combining our technology expertise with Avitar's leadership in the rapid, oral fluid drugs-of-abuse market affords Immunomatrix another opportunity to provide the diagnostic community with novel, proprietary technology," said Allan M. Weinstein, M.D., Chairman & Chief Executive Officer of Immunomatrix. "We look forward to working with the Avitar team in this development project."
Avitar, Inc. develops, manufactures and markets innovative and proprietary products in the oral fluid diagnostic market, disease and clinical testing market, and customized polyurethane applications used in the wound dressing industry. Oral fluid diagnostics include the estimated $1.5 billion drugs-of- abuse testing market, which encompasses the corporate workplace and criminal justice markets. Avitar's products include ORALscreen(TM), the world's first non-invasive, rapid, onsite oral fluid test for drugs-of-abuse, and HAIRscreen(TM), a laboratory-based hair test for detecting long-term drug abuse. Additionally, Avitar manufactures and markets HYDRASORB(TM), an absorbent topical dressing for moderate to heavy exudating wounds. For more information, see Avitar's website at www.avitarinc.com.

دورة المؤشرنت للتحليل الفني
50 دينار كويتي